Thanks for watching! Part 1: https://www.youtube.com/watch?v=kWUho_Cmv5s
Timestamps:
0:00 Leonhard Euler, Euler’s Identity
1:15 Rudolf Clausius, Second Law of Thermodynamics
2:38 Marie Curie, Law of Radioactive Decay
3:55 Daniel Bernoulli, Bernoulli’s Equation
4:52 Ludwig Boltzmann, Entropy Equation
6:04 James Joule, First Law of Thermodynamics
7:09 Georg Ohm, Ohm’s Law
8:01 Johannes Kepler, Third Law of Planetary Motion
9:15 Gaspard-Gustave Coriolis, Coriolis Force
Leonard Euler – Euler’s identity
Leonard Euler is responsible for one of the most admired formulas in the history of mathematics. Euler’s identity. This iconic equation connects five of the most fundamental numbers in mathematics. With three basic operations in a single expression;
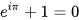
Where e is the base of the natural logarithm. I is the imaginary unit equivalent to i^2=1. Pi is the fundamental constant of trigonometry and 1 and zero are the neutral elements of multiplication and addition respectively.
This identity arises from Euler’s formula for complex exponential functions published in 1748 in his work introducio in analysis infinatorum.
Euler ‘s identity is a natural consequence of complex analysis which in that century was just beginning to take shape. Its elegance has led it to be regarded as a jewel of mathematical thought. Considered a perfect expression of mathematical harmony, this equation further cemented Euler’s status as a legend in the history of science.
Rudolf Clausius – Second Law of Thermodynamics

In 1850, German physicist Rudolph Clausius formulated one of the most important principles of physics, the second law of thermodynamics. This law describes the natural direction of thermodynamic processes and states that the entropy of a closed system always tends to increase.
In 1865, Clausius first coined the concept of entropy, introducing the equation
delta S is greater than or equal to Q / T

where delta S represents the entropy change, delta Q is the heat exchanged and T is the temperature in Kelvin.
This inequality indicates that disorder or irreversibility in a system never decreases spontaneously. For example, if you drop a cup and it breaks, the pieces don’t reassemble themselves. The system went from an ordered state, the whole cup, to a disordered one, the fragments. This transformation occurs spontaneously. But the reverse process, the pieces reassembling themselves, doesn’t happen without external intervention.
This is what means that entropy doesn’t decrease spontaneously. Clausius’s formulation laid the foundations of modern thermodynamics and profoundly influenced fields such as statistical physics, engineering, and cosmology. His work established him as one of the great pillars of 19th century physics.
Marie Curie – Law of Radioactive Decay

At the beginning of the 20th century, Marie Curie revolutionized physics by investigating radioactivity, a term she coined herself. Together with Pierre Curie, she discovered the elements polonium and radium in 1898 and formulated a key law for understanding radioactive decay.
Radioactive nuclei are inherently unstable and over time spontaneously decay to become more stable. The law of radioactive decay allows us to predict how many of these nuclei will remain undecayed after a certain time.
N of t= n * e to the negative lambda t

where n of t is the number of nuclei at time t
n the initial number and
lambda the decay constant.
This formula is fundamental in nuclear physics, medicine, archaeology and geology and the latter because it is the basis of the radiocarbon dating method also known as the carbon 14 test.
Marie Curie received two Nobel prizes in physics 1903 and chemistry 1911, becoming an icon of modern science. Her work opened new frontiers for atomic and medical research.
Daniel Bernoulli – Bernoulli’s Equation
In 1738, Daniel Bernoulli published Hydrodynamica, sive de Viribus et Motibus Fluidorum Commentarii, where he formulated the equation that bears his name and which became the cornerstone of fluid mechanics. It describes how energy is conserved in a moving fluid.
Bernoulli’s equation:
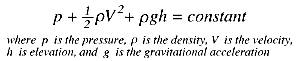
The formula expresses that the sum of pressure, kinetic energy and potential energy remains constant throughout an ideal fluid stream. Its application is crucial in aviation, medicine, hydraulic engineering, and vehicle design. This law allowed us to understand phenomena such as aircraft flight and blood flow. Elevating Bernoulli as a key figure in applied physics.
Ludwig Boltzmann – Entropy Equation
At the end of the 19th century, Ludwig Boltzmann explored the concept of entropy from a statistical perspective. His formula relates the microscopic disorder of a system to a macroscopically measurable quantity entropy.
The famous Boltzmann equation is:
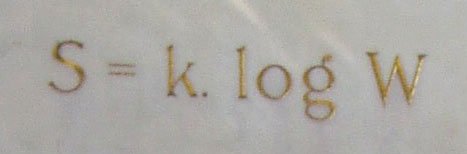
S= K * the logarithm of W.
S represents entropy.
K is the Boltzman constant and
W – Wahrscheinlichkeit (the German word for probability) of a macroscopic state for some probability distribution of possible microstates.
That is, W represents the number of different ways in which the internal parts of a system such as atoms or molecules can be organized without any difference being noticeable from the outside. In other words, they are all the possible combinations that give rise to the same general state of the system.
Boltzmann’s formula, even engraved on his tombstone, bridges the gap between classical mechanics and statistical theory. Although his theory was harshly criticized during his lifetime, his ideas became a cornerstone of modern physics.
His work explained why systems evolve toward disorder and why time has direction fundamental concepts in both thermodynamics and cosmology.
James Juul – first law of thermodynamics
In the 1840s, James Prescott Juel experimentally demonstrated that heat is not a substance but a form of energy. This idea was fundamental to establishing the first law of thermodynamics. The conservation of energy in thermodynamic systems. The law is expressed as;
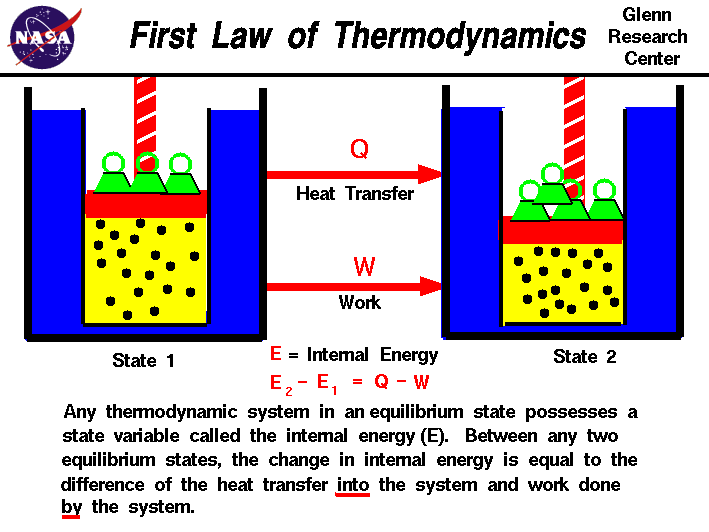
This formula unifies mechanics and thermodynamics under the principle of conservation of energy. In his experiment on the mechanical equivalence of heat, James Juul used a system in which a descending weight rotated paddles immersed in water by measuring the temperature increase. He demonstrated that mechanical work could be directly transformed into heat. This discovery helped consolidate a unified view of energy in the universe and laid the foundations of modern physics. The unit of energy the jewel is named in his honor.
Georg Ohm – Ohm’s law
In 1827, German physicist Georg Simon Ohm published “Die galvanische Kette, mathematisch bearbeitet” in which he formulated a quantitative relationship between voltage current and electrical resistance known as Ohm’s law.
The mathematical expression is V= IIR
Where V is voltage I is current and R is resistance.
This equation describes how electricity behaves in linear circuits laying the foundation for modern electronics. At first, his ideas were rejected by German academia. But over time, their crucial importance was recognized. Today, Ohm’s law is one of the most widely used principles in electrical and electronic engineering.
The Ohm (Ω), the unit of electrical resistance, is named after him in honor of his contribution to scientific and technological knowledge.
Johannes Kepler – Third Law of Planetary Motion
In 1619, Johannes Kepler formulated his third law of planetary motion in his work Harmonices Mundi. This law describes a precise mathematical relationship between a planet’s orbital period and its mean distance from the sun.
Kepler’s third law states for planets or satellites in a circular orbit about the same central body, the square of the time period is proportional to the cube of the radius of the orbit. The equation summarizing this law is

Where t is the orbital period, i.e the time it takes for a planet to go around the sun, and
r is the mean radius of the orbit.
This implies that planets farther from the sun move slower in wider orbits. Now, proportional does not exactly mean equal, but what it does mean is that we’re able to compare planet orbits by delving deeper with this equation. Kepler’s third law consolidated the heliocentric model and anticipated Newton’s laws of motion. It represented a milestone in the transition from ancient astronomy to modern physics and opened the door to understanding the dynamics of the solar system from a rigorous mathematical perspective.
Gaspard Gustave Coriolis – Coriolis force
Coriolis force. In 1835, the French engineer and mathematician Gaspard Gustav Coriolis described an apparent force acting on moving objects within a rotating reference frame. This force explains why hurricanes rotate in opposite directions in the Earth’s hemispheres.
The mathematical expression of the coriolis force is f sub c = -2 m * omega cross v

where m is the mass of the object.
Omega is the angular velocity vector of the rotating system and
v is the velocity of the object.
The vector product indicates that the force is perpendicular to both omega and v. Although it was conceived for rotating machines, its application in geophysics, meteorology, and oceanography is essential. The Coriolis force helps us understand wind patterns, ocean currents, and ballistic trajectories. Its discovery made Coriolis a key figure in the dynamics of noninertial systems.
This article was generated from the video transcript of “Every Math Problem that Made Someone Famous (pt. 2)”.
Watch the full video above for more visual explanations and diagrams.


Leave a Reply