The Ancient Idea of Atoms
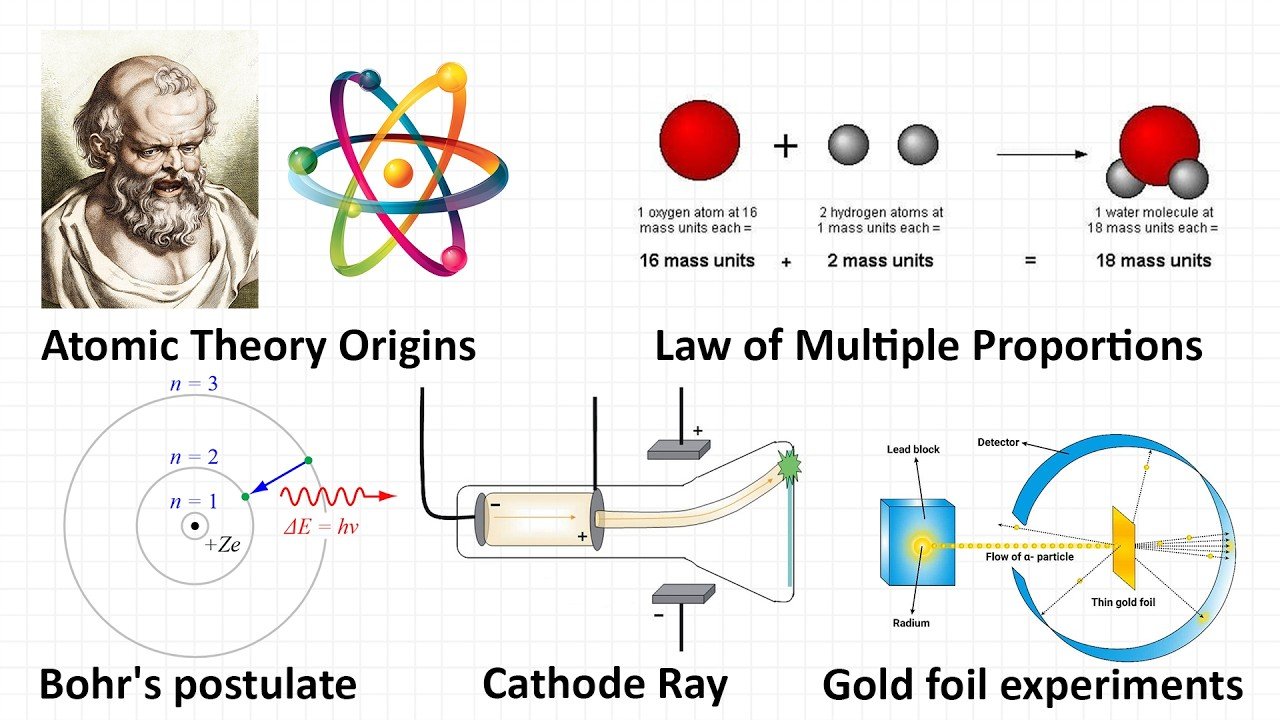
Our understanding of matter, everything we can touch, see, and feel, rests on the foundational idea that all things are made of atoms. This idea began in the fifth century BCE when Greek philosophers like Democritus proposed that everything was composed of small indivisible particles, which he called atomos.
Democritus believed that atoms are too small to be seen by the naked eye, that they are infinitely many, and that they have always existed. Atoms, Democritus argued, float in the void, and they are constantly moving and colliding with each other. Considering that Democritus lived thousands of years ago, during a time long before the invention of microscopes, he was very close to the truth.
But even though he was right about a lot of things, it would not be for nearly two millennia that his ideas were proved to be scientific fact.
John Dalton and Chemical Ratios
In the early 1800s, an English chemist called John Dalton noticed several intriguing patterns in chemical reactions. He was especially interested in how elements seem to combine in predictable whole number ratios.
A good example of this is the formation of water, which itself is a combination of oxygen and hydrogen. Dalton knew that the combination had to be in a fixed ratio by mass. For every 8 g of oxygen, only about 1 g of hydrogen was needed. This suggested that water was not a continuous substance, but was instead composed of discrete hydrogen and oxygen units combining in a definite ratio.
Dalton interpreted this as a reflection of the relative atomic masses of hydrogen and oxygen atoms and the simple way they come together. He also noticed a similar rule in nitric oxide and nitrogen dioxide. The masses of oxygen that combined with a fixed mass of nitrogen were in ratios such as 1:2.
This observation led Dalton to propose his law of multiple proportions. When two elements combine to form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.
The pattern of simple whole number ratios was a strong hint that matter was composed of individual countable entities, atoms.
J. J. Thomson and the Electron

In 1897, J. J. Thomson, a British physicist, ran a series of experiments with cathode rays. Initially, Thomson used a combination of magnets and electrometers, which proved that these rays carried an electric charge, but there was still the question of whether they were positively or negatively charged.
To answer that question, Thomson observed their behavior near charged metal plates. The rays clearly bent away from negatively charged plates, steering toward positive ones, confirming that they were negatively charged.
The real breakthrough moment came when Thomson measured their mass to charge ratio. By doing this, he discovered that cathode rays were not waves or some strange energy form, but tiny negatively charged particles. We now know them as electrons.
With this discovery, Thomson came up with what later writers would call the plum pudding model. He imagined the atom as a positively charged sphere studded with negatively charged electrons embedded throughout, a bit like raisins scattered within a plum pudding.
Rutherford and the Gold Foil Experiment

Since Dalton’s time, atoms were considered the smallest indivisible building blocks of matter. They were thought of as solid, indestructible spheres. But Thomson’s discovery of the electron showed that atoms had internal structure.
Even then, electrons were found to have extremely little mass compared to the whole atom. Scientists wondered what accounted for the bulk of an atom’s mass. That question was addressed by Ernest Rutherford and his colleagues Hans Geiger and Ernest Marsden in their famous gold foil experiments.
Rutherford directed beams of alpha particles at thin foils of metal like gold. Surrounding the foil was a screen coated with zinc sulfide, which made it fluorescent. Whenever an alpha particle struck the screen, it produced a tiny flash of light.
According to the plum pudding model, the positive charge of the atom was spread out, so alpha particles should have passed through with negligible deflection. Most of them did. But Rutherford noticed that a small number bounced off the foil at large angles.
In a lecture at Cambridge University, Rutherford described his shock. It was almost as incredible as if you fired a 15 inch shell at a piece of tissue paper and it came back and hit you.
These results led him to conclude that most of the atom is empty space. The deflected particles must have struck a very small, very dense, positively charged center. He called this central core the nucleus. From his data, he estimated that the nucleus was tens of thousands of times smaller than the atom as a whole, but contained nearly all its mass.
The Problem with Classical Physics
Rutherford’s atomic model had a serious flaw. Classical physics predicted that orbiting electrons should constantly emit electromagnetic radiation due to their acceleration. If that were true, electrons would lose energy and spiral into the nucleus. The atom would collapse within fractions of a second, which clearly does not happen.
At the same time, experiments with hydrogen gas revealed that atoms emit light at specific spectral lines rather than a continuous spectrum. Rutherford’s model could not explain this phenomenon.
Niels Bohr and Quantized Energy Levels

Another scientist, Niels Bohr, proposed a new idea. Inspired partly by Max Planck’s work on quantum theory, he suggested that electrons do not radiate energy when they are in certain special orbits.
He proposed that the angular momentum of the electron is quantized, given by the equation:
L = n h / 2π
In this equation, L is the angular momentum of an electron. The value n is a positive whole number, 1, 2, 3, and so on, known as the principal quantum number. It determines which orbit or energy level the electron is in. The constant h is Planck’s constant. The factor 2π arises from the circular nature of the orbit.
Electrons do not radiate energy when in these stable orbits. They only emit or absorb energy when jumping from one allowed orbit to another.
Bohr also used the relation:
E = h f
where E is the energy of the photon, h is Planck’s constant, and f is the frequency of the emitted light.
When Bohr applied this model to hydrogen, it explained the observed spectral lines in hydrogen’s emission spectrum. The allowed energy levels predicted by his model matched the Balmer series, the visible lines of light that hydrogen emits.
This explained why electrons do not spiral into the nucleus. An electron can only gain or lose energy by jumping between fixed orbits. When it jumps from a higher energy orbit to a lower one, it emits light with a frequency that matches the observed spectral lines.
Toward Quantum Mechanics
Even though Bohr’s model was a major advancement, it was incomplete. It successfully described the hydrogen atom but struggled to predict the behavior of more complex atoms.
The full explanation required the later development of quantum mechanics, with contributions from Erwin Schrödinger, Werner Heisenberg, and Paul Dirac.
Dalton, inspired by Democritus, argued that atoms were the smallest units of matter and that they combined in whole number ratios to form compounds. Thomson discovered electrons, showing that atoms were divisible. Rutherford discovered the dense nucleus at the center of the atom. Bohr refined the picture by explaining the quantized nature of electron energy levels.
Today, we understand atoms as complex structures composed of a dense nucleus surrounded by electrons described by quantum mechanics.
Further Reading
-
Stanford Encyclopedia of Philosophy, “Ancient Atomism”
-
Royal Society of Chemistry, “John Dalton and Atomic Theory”
-
Nobel Prize Biography of J. J. Thomson
-
Rutherford’s 1911 paper on nuclear structure
-
Niels Bohr, “On the Constitution of Atoms and Molecules” 1913
-
MIT OpenCourseWare, Introduction to Quantum Mechanics

Leave a Reply